Contents
ℹ️ NOTE
This measure has been removed as an eCQM for Traditional MIPS reporting.
It remains an applicable eCQM for other programs and will continue to be available in Elation. However, it will
not be eligible to count toward Traditional MIPS submissions via QRDA for the 2026 MIPS Performance Year. For a list of 2026 MIPS eligible eCQMs, see our
MIPS (2026) Overview article.
Measure Details
Percentage of adults 45-75 years of age who had appropriate screening for colorectal cancer
Measure Parameters
Patients with one or more screenings for colorectal cancer. Appropriate screenings are defined by any one of the following criteria:
- Fecal occult blood test (FOBT) during the measurement period.
- Flexible sigmoidoscopy during the measurement period or the four years prior to the measurement period.
- Colonoscopy during the measurement period or the nine years prior to the measurement period.
- Stool DNA (sDNA) with FIT test during the measurement period or the two years prior to the measurement period.
- CT Colonography during the measurement period or the four years prior to the measurement period.
Patients 46-75 years of age by the end of the measurement period with a visit during the measurement period.
- Exclude patients who are in hospice care for any part of the measurement period.
- Exclude patients with a diagnosis or past history of total colectomy or colorectal cancer.
- Exclude patients receiving palliative care during the measurement period.
- Exclude patients 66 and older by the end of the measurement period who are living long term in a nursing home any time on or before the end of the measurement period.
- Exclude patients 66 and older by the end of the measurement period with an indication of frailty for any part of the measurement period who also meet any of the following criteria advanced illness criteria:
- Advanced illness diagnosis during the measurement period or the year prior
- OR taking dementia medications for any part of the measurement period or the year prior
Elation Workflows
| 1 |
Click Address in the Colorectal Cancer Screening is due reminder at the top of the visit note draft.
|
| 2a |
To immediately meet the numerator requirements by documenting a Cologuard, FIT, FIT-DNA or FOBT test result for the patient
- Click Enter FOBT or Fit DNA result.
- Record the details of a Pap Smear or HPV Test:
- Enter the title of test.
- Enter the dates of the test in the Collected and Resulted fields.
- Enter the test results in the appropriate field.
- Click Sign or Sign & Export to Note to save the results.
|
| 2b |
To immediately meet the numerator requirements by linking a test result:
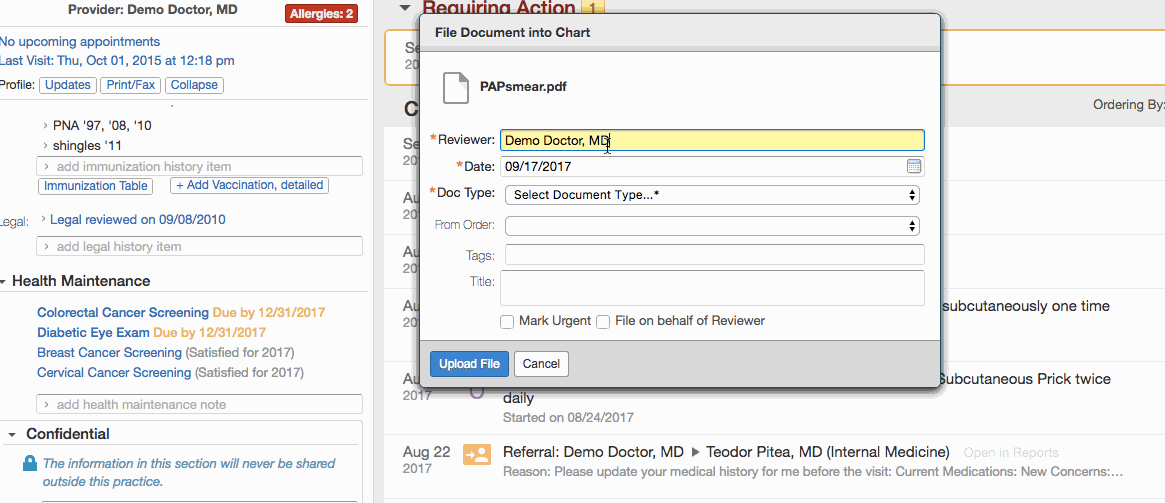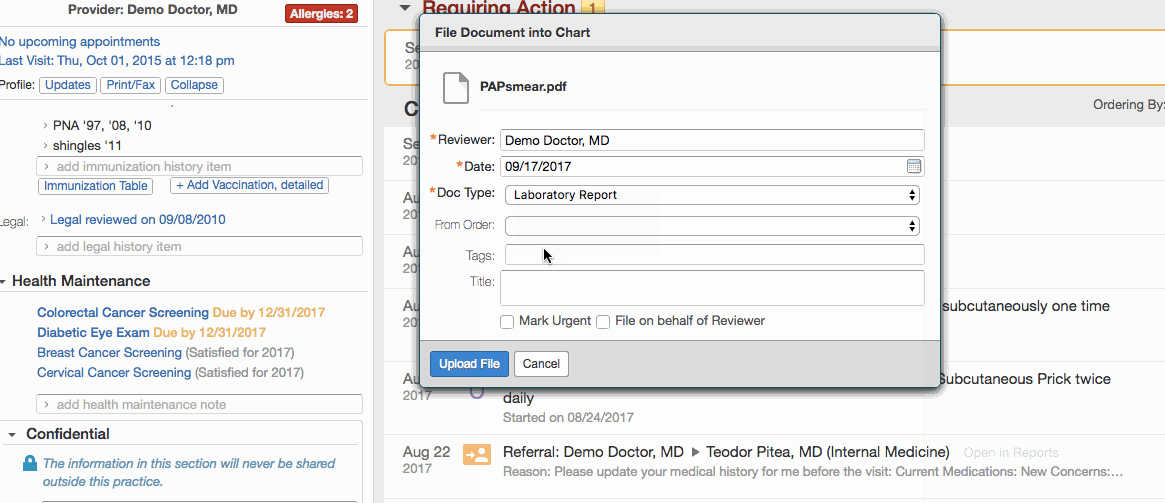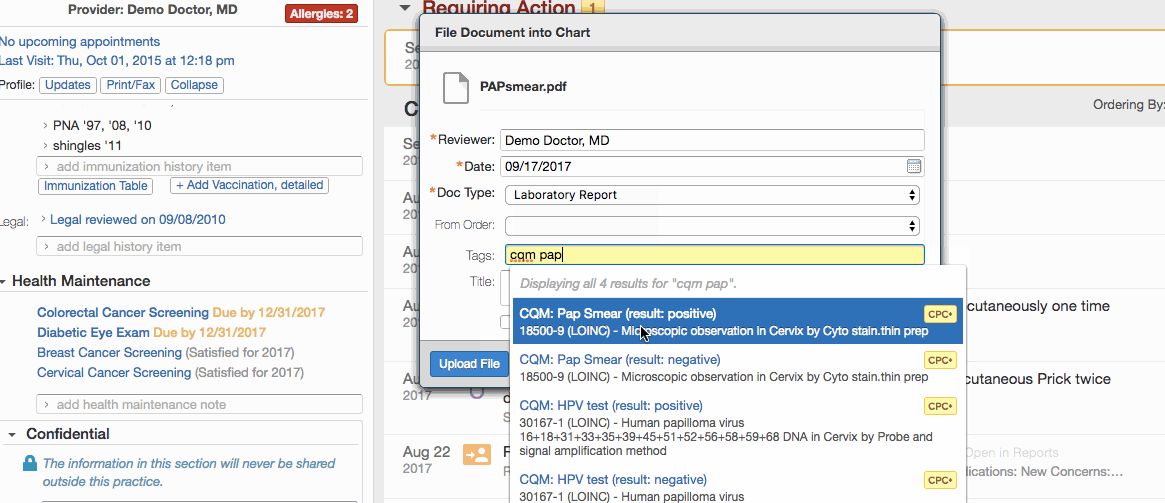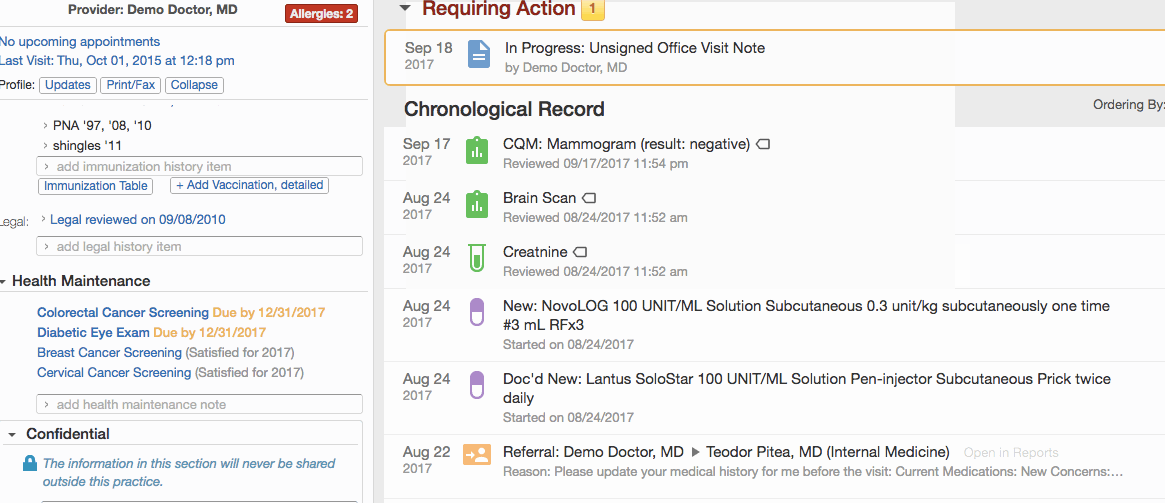
- Click Document existing screening results.
- Record the details of a test:
- Enter the date of the test in the Date field.
- Select the appropriate document tag in the Type field:
- COLONOSCOPY (RESULT: NEGATIVE)
- COLONOSCOPY (RESULT: POSITIVE)
- FIT DNA (RESULT: NEGATIVE)
- FIT DNA (RESULT: POSITIVE)
- FLEXIBLE SIGMOIDOSCOPY (RESULT: NEGATIVE)
- FLEXIBLE SIGMOIDOSCOPY (RESULT: POSITIVE)
- FOBT (RESULT: NEGATIVE)
- FOBT (RESULT: POSITIVE)
- CT COLONOGRAPHY (RESULT: NEGATIVE)
- CT COLONOGRAPHY (RESULT: POSITIVE)
- Click + Link a report to this item to link the test results in the patient's chart to the record.
- Click Save New.
|
| 2c |
To move closer to meeting the numerator requirements:
- Click Order FOBT or FIT DNA to create a lab order for the patient.
- Click Refer for colonoscopy to create a referral for the patient.
- Click Refer for CT colonography to create an imaging order for the patient.
- Click Refer for flexible sigmoidoscopy to create a referral for the patient.
- Click Send office message to follow-up on open order to have staff follow up on an outstanding lab order.
Once the test is completed and you have the results, return to Step 2a or 2b.
|
| 2d |
To exclude the patient due to total colectomy, click Document exclusion: patient had a total colectomy. This will append the EXCLUSION: COLECTOMY NO COL SCRN document tag to the visit note to exclude the patient from the measure.
Click here for instructions on how to document other exclusions/exceptions.
|
Clicking Dismiss will dismiss the reminder and the reminder will not appear again until a new visit note draft is opened.
| 1 | Click on Colorectal Cancer Screening in the Health Maintenance section of the Clinical Profile. |
| 2 | Enter the date of the test in the Date field. |
| 3 |
Select the appropriate document tag in the Type field:
- COLONOSCOPY (RESULT: NEGATIVE)
- COLONOSCOPY (RESULT: POSITIVE)
- FIT DNA (RESULT: NEGATIVE)
- FIT DNA (RESULT: POSITIVE)
- FLEXIBLE SIGMOIDOSCOPY (RESULT: NEGATIVE)
- FLEXIBLE SIGMOIDOSCOPY (RESULT: POSITIVE)
- FOBT (RESULT: NEGATIVE)
- FOBT (RESULT: POSITIVE)
- CT COLONOGRAPHY (RESULT: NEGATIVE)
- CT COLONOGRAPHY (RESULT: POSITIVE)
|
| 4 |
Click + Link a report to this item to link the test results in the patient's chart to the record.
|
| 5 |
Click Save New.
|

| 1 | Open the test results in the patient's chart. |
| 2 | Click Actions -> Edit Details. |
| 3 |
Go to the Tags field and select the appropriate document tag below:
- COLONOSCOPY (RESULT: NEGATIVE)
- COLONOSCOPY (RESULT: POSITIVE)
- FIT DNA (RESULT: NEGATIVE)
- FIT DNA (RESULT: POSITIVE)
- FLEXIBLE SIGMOIDOSCOPY (RESULT: NEGATIVE)
- FLEXIBLE SIGMOIDOSCOPY (RESULT: POSITIVE)
- FOBT (RESULT: NEGATIVE)
- FOBT (RESULT: POSITIVE)
- CT COLONOGRAPHY (RESULT: NEGATIVE)
- CT COLONOGRAPHY (RESULT: POSITIVE)
|
| 4 | Click Save. |
| Exclusion/Exception | Step(s) |
|
Patients whose hospice care overlaps the measurement period.
|
Click + Tag at the bottom of the visit note draft and add the appropriate document tag from this list:
- EXCLUSION: HOSPICE CARE
- EXCLUSION: IP DISCHARGE STATUS TO HEALTH CARE FACILITY FOR HOSPICE CARE
- EXCLUSION: IP DISCHARGE STATUS TO HOME FOR HOSPICE CARE
|
| Exclude patients with a diagnosis or past history of total colectomy or colorectal cancer. |
Add a diagnosis of total colectomy or colorectal cancer in the patient’s Problem List or visit note with a start date prior to December 31, 2026. Alternatively for total colectomy, you can add click + Tag at the bottom of the visit note draft and add the document tag - EXCLUSION: COLECTOMY NO COL SCRN
|
| Patients receiving palliative care for any part of the measurement period. | Click + Tag at the bottom of the visit note draft and add the following document tag - EXCLUSION: PALLIATIVE CARE |
|
Exclude patients 66 and older by the end of the measurement period who are living long term in a nursing home any time on or before the end of the measurement period.
| Click + Tag at the bottom of the visit note draft and add the following document tag - EXCLUSION: Living Long Term in a Nursing Home. |
|
Exclude patients 66 and older by the end of the measurement period with an indication of frailty for any part of the measurement period who also meet any of the following criteria advanced illness criteria:
- Advanced illness diagnosis during the measurement period or the year prior
- OR taking dementia medications for any part of the measurement period or the year prior
| See below. |
Documenting frailty
To document frailty in Elation using
- Frailty diagnosis, advanced illness codes and/or medications
- Add a frailty diagnosis to the Problem List. You must select from one of the frailty codes listed here.
- Record one of the following:
- an advanced illness diagnosis in 2 outpatient encounters using one of the codes listed here.
- an advanced illness in 1 inpatient counter using one of the codes listed here.
- a dementia medication in the patient's chart.
- A frailty device code
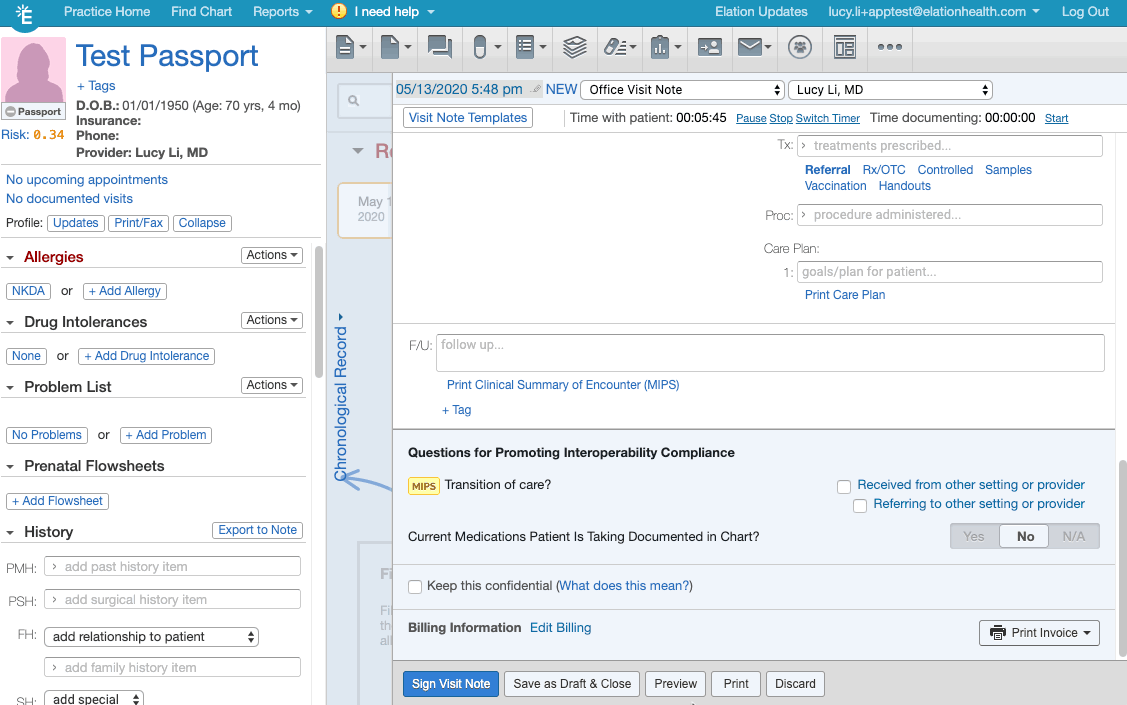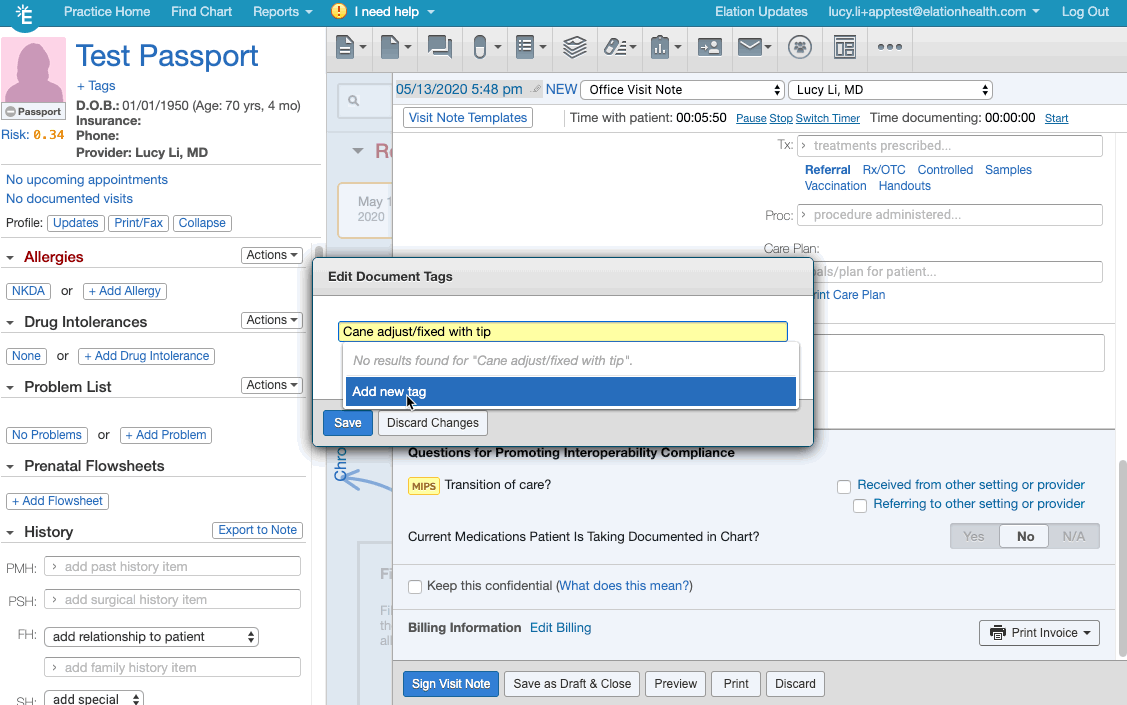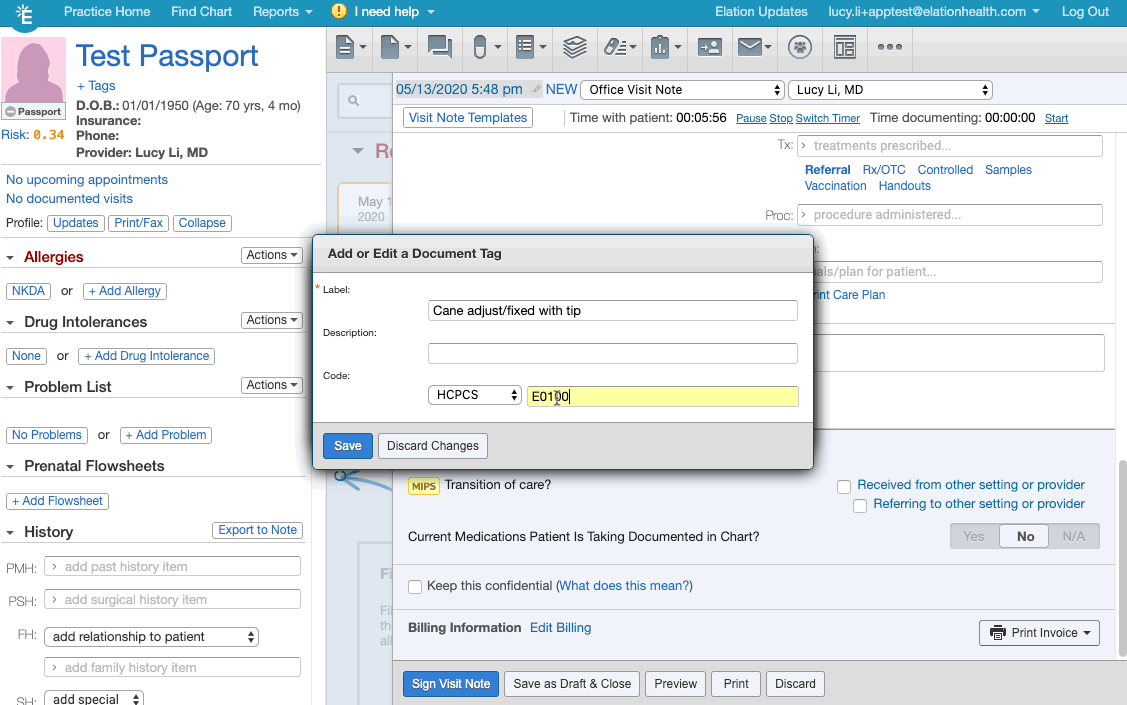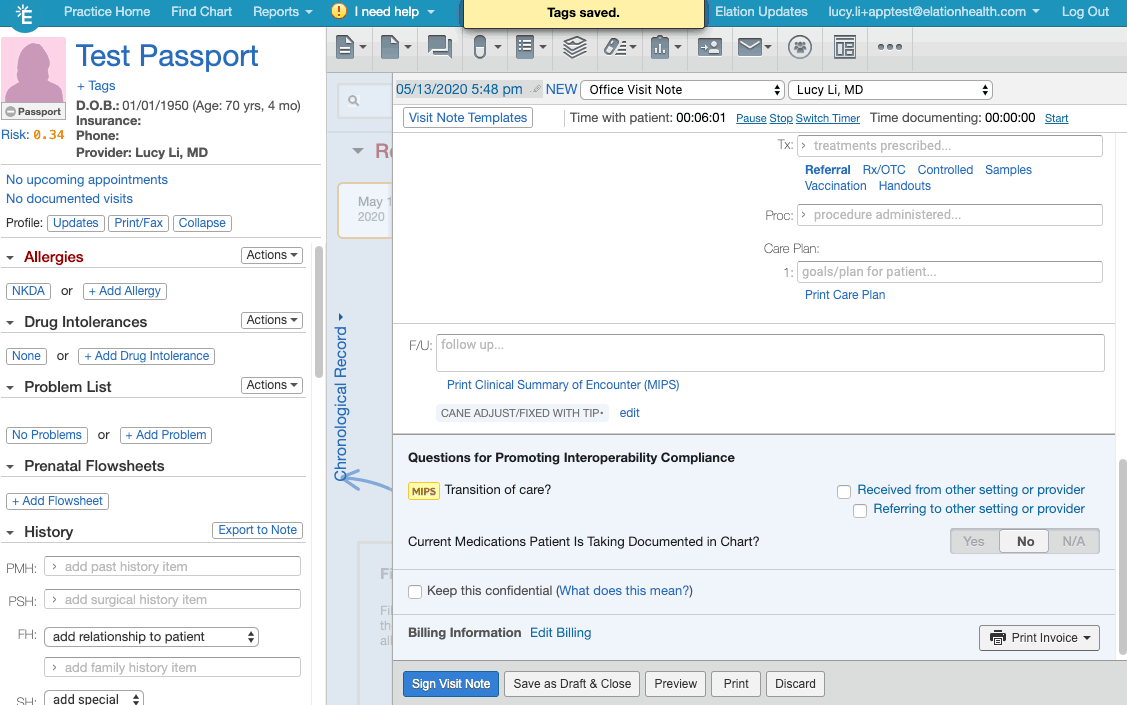
- Click + Tag at the bottom of the visit note draft.
- Create/add the appropriate document tag for the relevant frailty device from this list of frailty device codes.
- If you are creating the document tag for the first time for a frailty device, make sure the document tag has the device's HCPCS code listed.
Measure Information
Colorectal cancer represents eight percent of all new cancer cases in the United States. In 2020, there were an estimated 147,950 new cases of colorectal cancer and an estimated 53,200 deaths attributed to it. According to the National Cancer Institute, about 4.2 percent of men and women will be diagnosed with colorectal cancer at some point during their lifetimes. For most adults, older age is the most important risk factor for colorectal cancer, although being male and black are also associated with higher incidence and mortality. Colorectal cancer is most frequently diagnosed among people 65 to 74 years old (National Cancer Institute, 2020).
Screening can be effective for finding precancerous lesions (polyps) that could later become malignant, and for detecting early cancers that can be more easily and effectively treated. Precancerous polyps usually take about 10 to 15 years to develop into colorectal cancer, and most can be found and removed before turning into cancer. The five-year relative survival rate for people whose colorectal cancer is found in the early stage before it has spread is about 90 percent (American Cancer Society, 2020).
Reference: Measure Information from CMS
Attachments
Related Articles



